The Difference Between Governance Infrastructure and Governance Theater Most organizations don’t lack governance. They ...
Standard Operating Procedures - A complete guide
Standard Operating Procedures (SOP) are key to many businesses these days. Not only do they document the successful process to maintain a quality product, output or procedure. In many circumstances they are the key to ensuring regulatory compliance. This SOP guide provides an overview of the creation process.
The creation of a SOP is just one small part of success. The whole lifecycle needs to be considered from creation, through to training and distribution. Then, there needs to be consideration for review and updates during the SOP lifespan. In this series of blog posts, we'll delve into the crucial elements of SOPs, highlighting key areas to focus on. We'll also examine considerations related to technology, personnel, and processes for each major component.
You can also download this entire series of blog posts as an eBook.
What is an SOP?
Firstly, let's define what a SOP is. A SOP is a document that outlines the steps that are required to complete a particular process. SOPs are used in many different industries to ensure that work is done in a consistent and efficient manner. They can be used to document procedures for anything from manufacturing processes to customer service protocols. SOPs can help to improve the quality of work, reduce errors, and increase efficiency by providing clear and detailed instructions for completing tasks.
The SOP pie diagram
To aid our discussion, we've created the 'SOP Pie,' a high-level diagram that illustrates the SOP lifecycle and serves as a comprehensive guide to standard operating procedures.
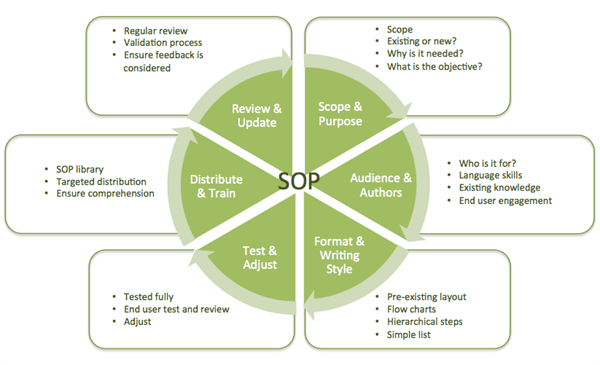
In truth there is no such thing as a ‘complete guide’ to SOPs, as the scenarios in which SOP’s are used are almost endless, and every situation has too many different variables to be able to list them all. There are however some quite simple considerations, checks and steps to help ensure success.
In each instalment of this guide, we will dig deeper into one of the six ‘slices’ in our ‘SOP pie’. We hope to provide a good insight in order to help ensure you and your organization are successful and efficient when it comes to SOPs.
Don’t miss our next blog which will cover the ‘Scope and Purpose’ considerations of the SOP lifecycle!... All of the posts in this series are linked below so you can easily navigate through them:
Get your free Standard Operating Procedures guide
Creating Standard Operating Procedures for your organisation doesn't have to be complicated. This guide will introduce you to the whole lifecycle from creation to training and distribution.
Other articles in the series:
You may also like:
The Difference Between Governance Infrastructure and Governance Theater Most organizations don’t lack governance. They ...
The Difference Between Governance Infrastructure and Governance Theater Most organizations don’t lack governance. They ...
The Difference Between Governance Infrastructure and Governance Theater Most organizations don’t lack governance. They ...
The Equifax Catastrophe: How One Missed Update Became a $700M Fine In most workplaces, ...
The Difference Between Governance Infrastructure and Governance Theater Most organizations don’t lack governance. They ...

